The reaction of 44.1 g of Cr203 with 35.0 g of Al produced 25.6 g of Cr. What is the percent yield for this reaction?
2Al + Cr203 + Al203 + 2Cr
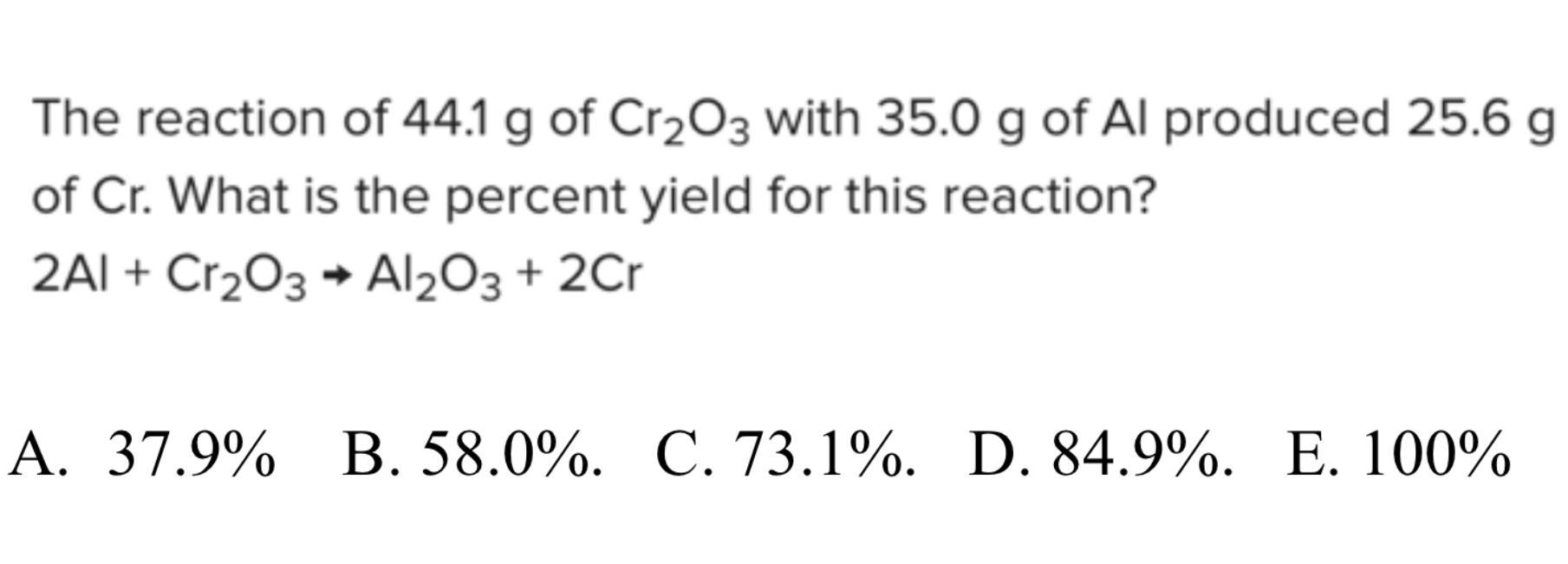
Answers
To determine the percent yield, we need to first calculate the theoretical yield of the reaction using stoichiometry, and then divide the actual yield by the theoretical yield and multiply by 100%. The percent yield of the reaction is approximately 84.9%.
What is percent yield?Percent yield is a measure of the efficiency of a chemical reaction, calculated by dividing the actual yield of a reaction by the theoretical yield and multiplying by 100%. It represents the percentage of the theoretical amount of product that was actually obtained in a reaction.
The balanced chemical equation is:
2Al + Cr₂O₃ → Al₂O₃ + 2Cr
The molar mass of Cr₂O₃ is 152 g/mol, the molar mass of Al is 27 g/mol, and the molar mass of Cr is 52 g/mol.
We need to determine which reactant is limiting, so we can calculate the theoretical yield based on the amount of limiting reactant. We can do this by calculating the number of moles of each reactant using their molar masses and dividing by their stoichiometric coefficients in the balanced equation:
moles of Cr₂O₃= 44.1 g / 152 g/mol = 0.29 mol
moles of Al = 35.0 g / 27 g/mol = 1.30 mol
From the balanced equation, we see that 1 mole of Cr2O3 reacts with 2 moles of Cr. Therefore, the theoretical yield of Cr is:
moles of Cr produced = 0.29 mol Cr₂O₃x (2 mol Cr / 1 mol Cr₂O₃) = 0.58 mol Cr
mass of Cr produced = 0.58 mol Cr x 52 g/mol = 30.16 g Cr
The percent yield is:
% yield = (actual yield / theoretical yield) x 100%
% yield = (25.6 g Cr / 30.16 g Cr) x 100% = 84.9%
Therefore, the percent yield of the reaction is approximately 84.9%.
To find out more about percent yield, visit:
https://brainly.com/question/17042787
#SPJ1
Related Questions
if a second-order reaction has a half-life of 10.0 minutes when the initial reactant concentration is 0.250 m, what is the half-life when the initial concentration is 0.050 m?
Answers
The half-life of the reaction with an initial concentration of 0.050 m is 16.9 minutes,
which is longer than the half-life of 10.0 minutes when the initial concentration was 0.250 m.
The half-life of a second-order reaction depends on the initial reactant concentration.
When the initial concentration of a reactant is higher, the half-life of the reaction will be shorter; when the initial concentration of a reactant is lower, the half-life of the reaction will be longer.
Therefore, if a second-order reaction has a half-life of 10.0 minutes when the initial reactant concentration is 0.250 m, the half-life when the initial concentration is 0.050 m would be longer than 10.0 minutes.
To determine the exact half-life of the reaction with the lower initial concentration, we can use the integrated rate law for a second-order reaction:
ln[A]t = -kt + ln[A]0
In this equation, A
is the initial concentration of the reactant; and k is the reaction rate constant.
The half-life of the reaction with an initial concentration of 0.050 m, we can rearrange the equation to solve for t, the time in which the reactant concentration decreases to half of the initial concentration:
t = -(1/k) ln[0.5A0]
The initial concentration of 0.050 m, solve for t to get the half-life of the reaction with the lower initial concentration:
t = -(1/k) ln[0.5(0.050)] = 16.9 minutes
Therefore, the half-life of the reaction with an initial concentration of 0.050 m is 16.9 minutes, which is longer than the half-life of 10.0 minutes when the initial concentration was 0.250 m.
to know more about half-life refer here:
https://brainly.com/question/24710827#
#SPJ11
a piece of metal with a mass of 31.5g is added to a graduated cylinder to calculate the volume. the water is initially at the 51 mark, and it rises to the 78 mark after the metal is added. what is the density of the metal?
Answers
The density of the metal is 1.167 g/ml.
The density of the metal can be calculated using the formula for density, ρ:
ρ = m /v
where ρ is the density, m is the mass, and v is the volume.
In this case, the mass of the metal is 31.5g and the volume can be determined by subtracting the initial volume (51mL) from the final volume (78mL) of water in the graduated cylinder. Thus, the volume of the metal is 27mL.
Using the formula, the density of the metal is then:
ρ = 31.5 g / 27mL
ρ = 1.167 g/ml
This means that 1 mL of the metal has a mass of 1.167g. Density is an important property of materials, as it affects other properties such as buoyancy. Generally, materials with a higher density will sink in a liquid, while those with a lower density will float.
Learn more about density here: https://brainly.com/question/1354972.
#SPJ11
a metal will be placed in fire and an electron will absorb enough energy to be promoted to a higher energy state. what do we call this higher energy state?
Answers
When a metal is placed in the fire and an electron absorbs enough energy to be promoted to a higher energy state, this higher energy state is referred to as the excited state.
An excited state is a state of a molecule or atom in which it has absorbed sufficient energy to move an electron from its current orbital to a higher orbital. This state is referred to as the excited state, and the electron that has been elevated to a higher energy level is said to be in an excited state.
The reason behind the electron's promotion to a higher energy state when a metal is placed in fire is that the heat causes the electrons to absorb energy, which causes them to move to a higher energy state. When electrons move to higher energy states, they release energy in the form of light, heat, or other radiation.
Learn more about electron https://brainly.com/question/26084288
#SPJ11
What is the pH of the solution obtained by mixing 35.00 mL of 0.250 M HCl and 35.00 mL of 0.125 M NaOH?
Answers
The pH of the solution obtained by mixing 35.00 mL of 0.250 M HCl and 35.00 mL of 0.125 M NaOH can be calculated as follows:
Let's understand this step-by-step:
1. HCl is an acid, while NaOH is a base. When an acid and a base react, they undergo a neutralization reaction, forming salt and water. The balanced chemical equation for the reaction between HCl and NaOH is:
HCl + NaOH → NaCl + H2O
This equation shows that 1 mole of HCl reacts with 1 mole of NaOH to produce 1 mole of NaCl and 1 mole of water.
Using the volumes and concentrations given in the question, we can calculate the moles of HCl and NaOH as follows: moles of HCl = 35.00 mL × 0.250 mol/L = 0.00875 mol
moles of NaOH = 35.00 mL × 0.125 mol/L = 0.004375 mol
The reaction between HCl and NaOH is 1:1, so the limiting reactant is NaOH because it has fewer moles. Therefore, all the NaOH will be used up, leaving some HCl unreacted. The number of moles of HCl that remain after the reaction is equal to the initial number of moles of HCl minus the number of moles of NaOH used up:
mol of HCl remaining = 0.00875 mol - 0.004375 mol = 0.004375 mol
The total volume of the solution is the sum of the volumes of the acid and the base:
Vtotal = Vacid + Vbase
Vtotal = 35.00 mL + 35.00 mL = 70.00 mL = 0.07000 L
The concentration of HCl in the solution is calculated using the number of moles of HCl remaining and the total volume of the solution:
[HCl] = mol of HCl remaining / Vtotal
[HCl] = 0.004375 mol / 0.07000 L
[HCl] = 0.0625 M
The pH of the solution can be calculated using the equation:
pH = -log[H+]
The concentration of H+ in the solution is equal to the concentration of HCl, so:
[H+] = [HCl] = 0.0625 M
Substituting this value into the pH equation:
pH = -log[H+]pH = -log(0.0625)pH = 1.20Therefore, the pH of the solution obtained by mixing 35.00 mL of 0.250 M HCl and 35.00 mL of 0.125 M NaOH is 1.20.
Learn more about pH: What is the approximate ph of a solution if the concentration of hydrogen ions is 5. 0 × 10–4 moles per liter? https://brainly.com/question/26424076
#SPJ11
a saturated hydrocarbon has the maximum amount of hydrogens attached to the carbon skeleton. group of answer choices true false
Answers
True, a saturated hydrocarbon has the maximum amount of hydrogens attached to the carbon skeleton.
What is a hydrocarbon?Hydrocarbons are organic molecules that are made up of only carbon and hydrogen atoms. They may be composed of chains of various lengths, rings of various sizes, or a combination of both. The simplest hydrocarbons, such as methane (CH4), ethane (C2H6), and propane (C3H8), are gaseous at room temperature, whereas larger hydrocarbons are liquids, such as hexane (C6H14), or solids, such as hexadecane (C16H34).
Unsaturated hydrocarbons have carbon-carbon double or triple bonds in their structures, indicating that they are not completely saturated with hydrogen atoms. These hydrocarbons are commonly referred to as alkenes or alkynes, respectively. Alkenes have one double bond, whereas alkynes have one triple bond.
Read more about hydrocarbon :
https://brainly.com/question/22047391
#SPJ11
the empirical formula of a chemical substance is ch2. the molar mass of a molecule of the substance is 56.108 g/mol. what is the molecular formula of the chemical substance? (4 points) c3h4 c4h8 c2h4 c6h6
Answers
The molecular formula of the chemical substance is C4H8.
The empirical formula of a chemical substance, CH2, and its molar mass of 56.108 g/mol can be used to calculate the molecular formula of the substance.
In order to do this, we need to divide the molar mass by the empirical formula mass. The empirical formula mass for CH2 is 12.011 g/mol, so the calculation is: 56.108 g/mol / 12.011 g/mol = 4.67.
4.67, is the ratio of the molecular mass to the empirical formula mass.
This means that the molecular formula of the chemical substance is C4H8, which has a molecular mass of 4 x 12.011 g/mol = 48.044 g/mol, and is the closest molecular mass to the given molar mass of 56.108 g/mol.
Therefore, the molecular formula of the chemical substance is C4H8.
to know more about molecular formula refer here:
https://brainly.com/question/14425592#
#SPJ11
what is the ph for a titration of 25.0 ml of 25.0 ml of 0.365 m acetic acid 0.365 m acetic acid when 10.3 ml of 0.432 m when 10.3 ml of 0.432 m naoh have been added?
Answers
The pH for a titration of 25.0 mL of 0.365 M acetic acid when 10.3 mL of 0.432 M NaOH have been added is approximately 4.69.
The pH for a titration of 25.0 mL of 0.365 M acetic acid when 10.3 mL of 0.432 M NaOH have been added can be calculated using the following steps:
1. Calculate the moles of acetic acid (CH₃COOH) and sodium hydroxide (NaOH) before the reaction:
- Moles of CH₃COOH = volume × concentration
= 25.0 mL × 0.365 mol/L
= 9.125 mmol
- Moles of NaOH = volume × concentration
= 10.3 mL × 0.432 mol/L = 4.4456 mmol
2. Determine the moles of acetic acid and sodium hydroxide remaining after the reaction: Since acetic acid and sodium hydroxide react in a 1:1 ratio, the limiting reactant will be NaOH.
- Moles of CH₃COOH remaining = 9.125 mmol - 4.4456 mmol = 4.6794 mmol - Moles of NaOH remaining = 0 mmol (all NaOH is consumed in the reaction)
3. Calculate the concentration of acetic acid and acetate ion (CH₃COO-) after the reaction:
- [CH₃COOH] = moles of CH₃COOH remaining / total volume
= 4.6794 mmol / (25.0 mL + 10.3 mL)
= 0.12998 mol/L
- [CH₃COO-] = moles of NaOH consumed / total volume
= 4.4456 mmol / (25.0 mL + 10.3 mL)
= 0.12346 mol/L
4. Calculate the pH using the Henderson-Hasselbalch equation:
pH = pKa + log([CH₃COO-] / [CH₃COOH]) pKa of acetic acid is 4.76, so:
pH = 4.76 + log(0.12346 / 0.12998) ≈ 4.69
Therefore, the pH for a titration of 25.0 mL of 0.365 M acetic acid when 10.3 mL of 0.432 M NaOH have been added is approximately 4.69.
Learn more about titration here:
https://brainly.com/question/2728613#
#SPJ11
does the hydrogen necessary in the electron transport chain come from the splitting of carbon dioxide molecules
Answers
The hydrogen necessary for this process is ultimately derived from the splitting of carbon dioxide molecules. Yes, the hydrogen necessary for the electron transport chain is derived from the splitting of carbon dioxide molecules in a process known as the Calvin Cycle, or the light-dependent reaction.
In this process, carbon dioxide, water, and light energy are used to create high-energy molecules, such as ATP and NADPH, which are then used in the electron transport chain. During the Calvin cycle, carbon dioxide is reduced by NADPH and ATP to produce a three-carbon molecule called glycerate 3-phosphate.
Hydrogen is removed from glycerate 3-phosphate to create a two-carbon compound known as glyceraldehyde 3-phosphate. This compound is then used to create other compounds, such as glucose, which can be used for energy.
Know more about electron transport chain here:
https://brainly.com/question/24372542
#SPJ11
a mixture of three gases has a total pressure of 1,380 mmhg at 298 k. the mixture is analyzed and is found to contain 1.27 mol co2, 3.04 mol co, and 1.50 mol ar. what is the partial pressure of ar? multiple choice 0.258 atm 301 mmhg 356 mmhg 5,345 mmhg 8,020 mmhg
Answers
The partial pressure of Ar is 0.219 * 1,380 mmHg = 301 mmHg.
The partial pressure of a gas in a mixture is equal to the mole fraction of that gas times the total pressure of the mixture.
The mole fraction of Ar in this mixture is 1.50/6.81 = 0.219. Thus, the partial pressure of Ar is 0.219 * 1,380 mmHg = 301 mmHg.
The ideal gas law states that the pressure of a gas is directly proportional to its number of moles and inversely proportional to its volume.
This law is expressed in the equation PV = nRT, where P is the pressure, V is the volume, n is the number of moles, R is the ideal gas constant, and T is the temperature.
In a mixture of gases, each gas behaves independently according to the ideal gas law. Thus, the total pressure of the mixture is the sum of the partial pressures of each gas.
The partial pressure of a gas is equal to its mole fraction times the total pressure. The mole fraction of a gas is the number of moles of that gas divided by the total number of moles of all gases in the mixture.
In the example provided, the total pressure of the mixture is 1,380 mmHg, the number of moles of CO2 is 1.27, the number of moles of CO is 3.04, and the number of moles of Ar is 1.50.
The total number of moles of all gases in the mixture is 1.27 + 3.04 + 1.50 = 6.81. The mole fraction of Ar is 1.50/6.81 = 0.219. Thus, the partial pressure of Ar is 0.219 * 1,380 mmHg = 301 mmHg.
To know more about partial pressure refer here:
https://brainly.com/question/13199169#
#SPJ11
How much KNO3 will dissolve in 200 grams H2O at 70 C
Answers
The red line shows that at 70 °C, 200 g of water will be saturated with about 140 g or potassium nitrate.
How does solubility in 100 grammes of water become calculated?This mass of a compound would be divided by mass of the solvent, and then divided by 100 g to determine its solubility. This calculation will give the solubility of the substance in g/100g.
How does the temperature affect KNO3's solubility in water?The curves demonstrate that when temperature rises, solubility of any and all three solutes increases. The most noticeable increase in solubility is for potassium nitrate, which goes from about 30 g per 100 g of water from over 200 grams per 100 grams of water.
To know more about potassium visit:
https://brainly.com/question/13321031
#SPJ1
A biochemist wanted to adjust the of of a buffer solution composed of ( ) and , by adding moles of. (assume the volume of the solution is not affected by this addition. ) determine the of the resulting solution:
Answers
The pH value of the resulting solution assume the volume of the solution is not affected by this addition is 3.283.
The pH scale determines how acidic or basic water is. The range is 0 to 14, with 7 representing neutrality. Acidity is indicated by pH values below 7, whereas baseness is shown by pH values above 7. In reality, pH is a measurement of the proportion of free hydrogen and hydroxyl ions in water.
In this Question, HF is a Weak Acid and RbF is a weak Base - HNO3 is a strong acid.
HF reaction in aqueous medium
HF + H2O --------- H3O+ + F -
RbF + H2O ---- Rb+ + F -
pH (Original) = pKa + log ( [salt ] / [Acid] )
We donot need to calculate pH original -which is for the original solution before adding the strong acid.
HF is a weak acid - so in a buffer solution its dissociation is negligible - so it does not affect the H+ ion concentration much.
When a 0.012 mol of HNO3 is added to the buffer solution , it dissociates in H+ and NO-3 .
H+ ions dissociated from the Acid react with F - and produce HF . As a result the acid concentration will increase to the extent of 0.012 mol and the salt concentration reduces by the same extent - 0.012 mol.
So the formula for New pH changes to
pH (New) = pKa + log ( [salt ] - 0.012 mol / [Acid] + 0.012 mol)
Here , 0.012 mol are added to 281 mL solution,
Concentration of HNO3, M = number of moles / Vol in litres
= 0.012 mol / 281 mL
= 0.012 mol / 281 / 1000
= [0.012 mol x 1000] / 281 L = 0.043 M
As pKa = -log(Ka) ,
Given [salt ] = 0.480 M , [Acid] = 0.318 M
= - log(Ka) + log [ (0.480 M - 0.043 M) / (0.318 M + 0.043 M) ]
= - log (6.31 x 10-4 ) + log ( 0.437 / 0.361)
pH (New) = 3.20 + 0.083 = 3.283.
Learn more about pH value:
https://brainly.com/question/10603602
#SPJ4
Complete question:
A biochemist wanted to adjust the pH of 281 mL of a buffer solution composed of 0.318 M HF and 0.480 M RbF (K, = 6.31e - 04) by adding 0.012 moles of HNO3. Determine the pH of the resulting solution: pH number (rtol=0.02, atol=1e-08)
The Quantum Theory Model seems to contradict one the above scientist's hypothesis. Who is it and why? Is there more than one?
Answers
Answer:
Multiple scientists, including Albert Einstein, David Bohm, John Bell, and Roger Penrose, have challenged certain aspects of quantum theory due to differing views about particle behavior, hidden variables, and consciousness. Despite the challenges, quantum theory remains widely accepted as one of the most accurate and well-tested frameworks in modern physics.
the volume of a container expands when it is heated from 159k to 456k. what was the original volume if the final volume is 15.5 l
Answers
The original volume of the container is 5.40 L.
The given final volume of a container when heated is 15.5 L. The container expands when heated from 159 K to 456 K.
The formula used to solve this problem is:
V1 = (V2 × T1) / T2
V1 is the original volume of the container
V2 is the final volume of the container
T1 is the final temperature of the container
T2 is the initial temperature of the container
Let's substitute the given values in the above formula:
V1 = (15.5 × 159) / 456V1 = 5.40 L
Therefore, the original volume of the container is 5.40 L.
To know more about original volume click here:
https://brainly.com/question/12014506
#SPJ11
a chemist determined by measurements that moles of magnesium participated in a chemical reaction. calculate the mass of magnesium that participated in the chemical reaction.
Answers
Answer : The molar mass of magnesium is 24.305 g/mol
To calculate the mass of magnesium that participated in the chemical reaction, you need to know the number of moles of magnesium and the molar mass of magnesium. The molar mass of magnesium is 24.305 g/mol. Multiply the number of moles of magnesium by the molar mass of magnesium to calculate the mass of magnesium that participated in the chemical reaction.
For example, if you were given that the number of moles of magnesium is 0.25 moles, then you can calculate the mass of magnesium by multiplying 0.25 moles by 24.305 g/mol. This gives a result of 6.076 g of magnesium that participated in the chemical reaction.
To sum up, calculating the mass of magnesium that participated in the chemical reaction requires knowing the number of moles of magnesium and the molar mass of magnesium. The molar mass of magnesium is 24.305 g/mol, and you can calculate the mass of magnesium that participated in the chemical reaction by multiplying the number of moles of magnesium by the molar mass of magnesium.
Know more about magnesium here:
https://brainly.com/question/1533548
#SPJ11
In a Lab session, you were asked to:
1. Model one of the chemical reaction types: Synthesis, Decomposition, or replacement.
2. List the elements/ compounds you used in your reaction.
3. Describe the reaction as endothermic or exothermic. Justify your answer.
4. Record a video demonstrating the modelling.
5. Explain how a closed system is suitable for your reaction. Relate your answer to law of conservation of mass.
6. During the reaction, the reactants had a potential energy of 400 KJ. As for the final products it had 200 KJ. Demonstrate the reaction by drawing the graph.
7. Identify if the reaction is an exothermic or endothermic reaction. Explain.
8. Interpret the factors that might affect your reaction rate.
Answers
1. I modeled a decomposition reaction.
2. used hydrogen peroxide (H2O2) as the compound for the reaction.
3. The reaction is exothermic. This is because the decomposition of hydrogen peroxide releases heat and energy, which can be observed through the effervescence or bubbling of the solution.
4. I recorded a video demonstrating the experiment and the resulting reaction.
5. A closed system is suitable for this reaction because it follows the law of conservation of mass, which states that mass cannot be created or destroyed, only transferred or transformed.
6. The potential energy diagram for this reaction would show the reactants at a higher energy level (400 KJ) and the products at a lower energy level (200 KJ), with the difference in energy being released as heat and energy.
7. The reaction is exothermic because it releases heat and energy, as observed through the effervescence or bubbling of the solution.
8. Factors that could affect the reaction rate include temperature, catalysts, and concentration of the reactants.
What is decomposition reaction?
A decomposition reaction is a type of chemical reaction in which a compound breaks down into two or more simpler substances. This type of reaction usually requires the addition of energy, such as heat or light, to break the bonds holding the compound together.
To know more about decomposition reaction, visit:
https://brainly.com/question/16987748
#SPJ1
does the response of temperature in the atmosphere to an increase in co2 always stay the same as the co2 is progressively increased?
Answers
Yes, the response of temperature in the atmosphere to an increase in CO2 is generally consistent. As more CO2 is added to the atmosphere, it traps more heat from the sun, leading to a gradual increase in temperature. This phenomenon is known as the greenhouse effect.
The response of temperature in the atmosphere to an increase in CO2 does not always stay the same as the CO2 is progressively increased. It changes depending on various factors. This statement is backed up by scientific evidence.CO2 is known as a greenhouse gas that warms the Earth's atmosphere by absorbing and radiating energy within the infrared range.
When there is more CO2 in the atmosphere, there will be more radiation absorbed and radiated, resulting in a temperature increase.
Therefore, as the concentration of CO2 rises, the temperature of the Earth's atmosphere should also rise. However, the relationship between CO2 and temperature is not that simple.
Read more about temperature :
https://brainly.com/question/24746268
#SPJ11
old ammunition or fireworks, lithium-sulfur batteries, wastes containing cyanides or sulfides, and chlorine bleach and ammonia are examples of which type of hazardous waste?
Answers
These are all examples of chemical hazardous waste. Chemical hazardous waste is waste that is flammable, reactive, corrosive, or toxic. It can include things like unused pesticides, paint, cleaning products, or batteries.
Old ammunition or fireworks, lithium-sulfur batteries, wastes containing cyanides or sulfides, and chlorine bleach and ammonia are examples of Household hazardous waste.What is hazardous waste?Hazardous waste is a waste material that is harmful to human health or the environment. Every year, households and businesses generate hazardous waste in various forms. Because hazardous waste may be flammable, poisonous, reactive, or corrosive, it requires special disposal procedures. Hazardous wastes must be properly disposed of to safeguard human health and the environment.Household hazardous waste (HHW) is the type of waste that can be found in a typical home. This waste is produced by households when they use products that contain harmful chemicals. Old ammunition or fireworks, lithium-sulfur batteries, wastes containing cyanides or sulfides, and chlorine bleach and ammonia are examples of household hazardous waste.
For more such questions on hazardous waste
https://brainly.com/question/4055432
#SPJ11
In order for a six-membered ring to undergo an E2 reaction, the substituents that are to be eliminated axial must both be in ___ positions When bromine and an adjacent hydrogen are both in axial positions, the large tent-butyl substituent is in an ____ position in the trans isomer. Because a large substituent is more stable in an ___ position than in an ____ position, elimination of the ___ isomer occurs through its more stable chair conformer, whereas elimination of the ___ isomer has to occur through its less stable chair conformer. The ____ isomer, therefore, reacts more rapidly in an E2 reaction
equatorial cis axial
trans
Answers
In order for a six-membered ring to undergo an E2 reaction, the substituents that are to be eliminated axially must both be in equatorial positions.
This is because when bromine and an adjacent hydrogen are both in axial positions, the large tent-butyl substituent is in a cis position in the trans isomer.
Because a large substituent is more stable in a cis position than in an axial position, elimination of the trans isomer occurs through its more stable chair conformer, while elimination of the cis isomer has to occur through its less stable chair conformer. The cis isomer, therefore, reacts more rapidly in an E2 reaction.
because the more stable conformer has to be destabilized in order for the reaction to proceed. As a result, the reaction rate is much higher for the trans isomer than for the cis isomer.
Know more about equatorial here
https://brainly.com/question/24168112#
#SPJ11
what is the percentage of the renantiomer in a sample of limonene that has a specific rotation ot -38, given that the specific rotatic of (s)-limonene is - 116?
Answers
Answer: The percentage of the (R)-limonene in the sample is 67.24%.
The percentage of the (R)-limonene in a sample of limonene with a specific rotation of -38 can be calculated using the following equation:
Percentage (R)-limonene = (Specific rotation of sample - Specific rotation of (S)-limonene) ÷ (Specific rotation of (S)-limonene) x 100%
In this case, the equation is:
Percentage (R)-limonene = (-38 - (-116)) ÷ (-116) x 100% = 67.24%
Therefore, the percentage of the (R)-limonene in the sample is 67.24%.
Learn more about enantiomer here:
https://brainly.com/question/30845514#
#SPJ11
i. if you conducted this coupling step under acidic conditions, how would you expect the reaction rate to be affected?
Answers
If you conducted this coupling step under acidic conditions, you expect the reaction rate to be affected because at low pH values, the carboxylic acid is transformed into a more electrophilic species, which is easily attacked by the nucleophile, and the yield of the amide bond would be high.
In organic synthesis, coupling reactions are common, and they include the combination of a nucleophile with an electrophile to form a covalent bond. The coupling reaction between a carboxylic acid and an amine is a straightforward way to synthesize an amide in the presence of an activating agent (a molecule that can increase the electrophilicity of the carboxylic acid).
It is worth noting that there are various methods for synthesizing amides, including chemical and enzymatic methods. Coupling reactions are the most frequent chemical methods used for the synthesis of amides.
Learn more about nucleophile at:
https://brainly.com/question/6357352
#SPJ11
citric acid contains 37.51% c, 4.20% h, and 58.29% o by mass. what is the empirical formula of this compound?
Answers
The empirical formula of the given compound can be determined as follows the CHOS or C3H4O3.
According to the given data, the compound citric acid contains 37.51% C, 4.20% H, and 58.29% O by mass. So, let's assume that we have 100 g of citric acid, and then, we can find the masses of each element present in it: Mass of carbon = 37.51 gMass of hydrogen = 4.20 g. Mass of oxygen = 58.29 g.
Next, we need to convert the masses into the number of moles using the molar masses of the elements. The molar mass of carbon = 12.01 g/mol, Number of moles of carbon = 37.51 g / 12.01 g/mol = 3.124 molMolar mass of hydrogen = 1.01 g/molNumber of moles of hydrogen = 4.20 g / 1.01 g/mol = 4.158 molMolar mass of oxygen = 16.00 g/molNumber of moles of oxygen = 58.29 g / 16.00 g/mol = 3.643 follow, we need to find the simplest whole-number ratio of these moles by dividing them by the smallest number of moles, which is 3.124 mol: Carbon = 3.124 mol / 3.124 mol = 1Hydrogen = 4.158 mol / 3.124 mol = 1.33 ≈ 1Oxygen = 3.643 mol / 3.124 mol = 1.17 ≈ 1So, the empirical formula of citric acid is CHOS or C3H4O3.
Read more about carbon:
https://brainly.com/question/19083306
#SPJ11
Does the phrase “Survival of the fittest” refer to an individual (single organism) or a species (group of same organisms)? Why?
Answers
The phrase "survival of the fittest," popularised in Charles Darwin's fifth edition of On the Origin of Species (published in 1869), argued that animals most adapted to their environment have the best chances of surviving.
What does "survival of the fittest" mean in terms of species?The environment and its conditions are continually changing, and the fittest individuals must generate even more fit offspring in order to ensure their survival. Here is when evolution comes into play.
Are organisms who are physically fitter more likely to survive and pass on their genes?An evolutionary mechanism is natural selection. Environment-adapted organisms have a higher chance of surviving and dispersing the genes that contributed to their success.
To know more about Charles Darwin's visit:-
brainly.com/question/13428382
#SPJ9
Consider the Baeyer permanganate test and chromic acid tests. These tests work by converting an aldehyde to what functional group? 1 KMnO4 and H2CrO4 act as what kind of reagent? (e.g. electrophile, nucleophile, oxidizing agent, reducing agent, acid catalyst, base catalyst, solvent etc.) 2. 3. Why does a ketone not react with these reagents?
Answers
The Baeyer permanganate test and chromic acid tests work by converting an aldehyde to a carboxylic acid functional group.
KMnO₄ and H₂CrO₄ act as oxidizing agents. A ketone does not react with these reagents because it does not have a hydrogen atom attached to the carbonyl group.
How does the Baeyer permanganate test work?The Baeyer permanganate test is used to identify the presence of unsaturation (i.e. double bonds) in a compound. When a double bond is present in the compound, it will be oxidized by potassium permanganate (KMnO₄) to form a diol functional group. In the case of aldehydes, the double bond is present between the carbonyl carbon and the hydrogen atom.
Therefore, the reaction will convert an aldehyde to a carboxylic acid functional group. This reaction is also known as the oxidation of aldehydes with KMnO₄.
What is the chromic acid test?The chromic acid test is another method for identifying the presence of unsaturation in a compound. It uses chromic acid (H₂CrO₄) as the oxidizing agent. Like the Baeyer permanganate test, the chromic acid test will convert an aldehyde to a carboxylic acid functional group. It is important to note that the chromic acid test is more sensitive to the presence of double bonds than the Baeyer permanganate test.
Therefore, it is often used as a confirmatory test after a positive result is obtained from the Baeyer permanganate test.
Learn more about chromic acid test on:
https://brainly.com/question/30824151
#SPJ11
with an atomic number of 11, which of these elements gets its symbol from the latin word natrium?
Answers
The element with an atomic number of 11 that gets its symbol from the Latin word "natrium" is Sodium. Its symbol is "Na".
The symbol for sodium is Na, which is derived from the Latin word natrium. Sodium is a soft, silvery-white, highly reactive metal that is a member of the alkali metal group. It is an important element for many biological processes and is commonly found in salt (sodium chloride).
The other elements listed in the question are chlorine, iron, and nitrogen. Chlorine has an atomic number of 17, iron has an atomic number of 26, and nitrogen has an atomic number of 7. None of these elements gets their symbol from the Latin word natrium.
For more question on atomic number click on
https://brainly.com/question/11353462
#SPJ11
Probable question would be
with an atomic number of 11, which of these elements gets its symbol from the latin word natrium?
Sodium
Chlorine
Iron
Nitrogen
molecules in which three atoms are arranged in a straight line are said to have ? geometry. quiz
Answers
Answer: Molecules in which three atoms are arranged in a straight line are said to have linear geometry.
What is a linear molecule?
A linear molecule is a molecule that has three or more atoms arranged in a straight line. Two main groups of linear molecules exist: homonuclear and heteronuclear. A homonuclear linear molecule has two or more identical atoms bonded to the central atom, whereas a heteronuclear linear molecule has two or more distinct atoms bonded to the central atom.
Examples of linear molecules include carbon dioxide (CO2), hydrogen cyanide (HCN), nitrogen dioxide (NO2), and sulfur dioxide (SO2).
Linear geometry is the shape of the molecule, which is governed by its geometry. The distribution of bonding electrons and non-bonding pairs in a molecule determines its shape. For instance, in a molecule with linear geometry, the bond angle between two atoms is 180 degrees (a straight line).
Learn more about linear molecule here:
https://brainly.com/question/16770400#
#SPJ11
how many moles of naoh will react with 0.50 mol of h2co3?
a. 0,25 mol NaOH
b. 0.50 mol NaOH
c. 1.0 mol NaOh
d. 2.0 mol NaOH
Answers
We will need 1.0 mol NaOH to react with 0.5 mol pf H2CO3.
Let's understand this in detail:
The balanced chemical equation of the neutralization reaction between H2CO3 and NaOH is
H2CO3 + 2NaOH ⟶ Na2CO3 + 2H2O.
We need to use the mole ratio from the balanced equation to determine how many moles of NaOH will react with 0.50 mol of H2CO3. We can see from the equation that 1 mole of H2CO3 reacts with 2 moles of NaOH.
Therefore, 0.50 mol of H2CO3 will react with
(2/1) x 0.50 = 1.0 mol of NaOH.
Answer: c. 1.0 mol NaOH.
#SPJ11
Learn more about neutralization reaction: Define Neutralisation reaction along with examples. https://brainly.com/question/23008798
describe how the orientaon of the glycosidic bond affects the properes of the polysaccharides it creates.
Answers
The orientation of the glycosidic bond affects the properties of the polysaccharides it creates by determining the geometry of the sugar units in the polymer chain. When the glycosidic bond is in the alpha configuration, the sugar ring has a twisted conformation, which results in the sugar units being oriented in a more linear fashion.
In contrast, when the glycosidic bond is in the beta configuration, the sugar ring has a more planar conformation, which results in the sugar units being oriented in a more zig-zag fashion.
This difference in orientation affects the overall structure of the polysaccharide. Polysaccharides with alpha glycosidic bonds tend to form helical structures, while polysaccharides with beta glycosidic bonds tend to form sheet-like structures. This is because the twisted conformation of the alpha sugar units allows for the formation of hydrogen bonds between adjacent sugar units, which leads to the formation of a helix.
In contrast, the more planar conformation of the beta sugar units does not allow for the formation of hydrogen bonds between adjacent sugar units, which leads to the formation of a sheet.
Additionally, the orientation of the glycosidic bond affects the solubility and digestibility of the polysaccharide. Polysaccharides with alpha glycosidic bonds tend to be more soluble and more easily digested than polysaccharides with beta glycosidic bonds.
This is because the helical structure of alpha-polysaccharides allows for more surface area to be exposed to water and digestive enzymes, while the sheet-like structure of beta-polysaccharides does not.
Learn more about polysaccharides here:
https://brainly.com/question/30529578#
#SPJ11
100cm3 of a gas at 27degree Celsius exert a pressure of its volume is increased to 200cm3 at 127 degrees Celsius
Answers
Answer: 100cm3 of gas at 27°c exert a pressure of 750mmHg. Calculate its pressure if it's volume is increased to 250cm3 at 127°c? In Chemistry
Explanation:
How many moles are in 3.5 moles of FeF3
Answers
We just use molar mass for FeF3 (129.9 g/mol) to calculate the number moles in 3.5grammes of FeF3. Hence, just 3.5 x 129.9 = 4546.5 moles of FeF3 need to be multiplied.
Describe the Mass.An object's mass is determined by how much matter it has. Something that has more substance will weigh heavier overall. For instance, because an elephant contains more stuff than a mouse does, it has a heavier mass.
55.8+3⋅19=116 g/mole24 g116 g/mol=0.207 moles of FeF3
0.207 moles×6.022×23molecules/mole=1.2×1023molecules
How is mass measured?A thing's mass is how much matter it contains. Using a balance, scientists frequently determine mass. A beam balance or perhaps an electronic balance can be used to measure the mass of solids directly. Measure a liquid's volume, then use the density table to determine the liquid's mass.
To know more about mass visit:
https://brainly.com/question/15959704
#SPJ1
what term refers to the ability of open systems to fight off deterioration, sustain themselves and grow? a. requisite variety b. network properties c. negative entropy d. modeling techniques
Answers
The ability of open systems to fight off deterioration, sustain themselves and grow is Negative Entropy. Correct answer is option C
Negative Entropy is an important concept in thermodynamics and physics, where it is defined as a decrease in the entropy of a system. Entropy is the measure of randomness or disorder in a system, so negative entropy indicates that a system is becoming more organized, or that it is moving away from equilibrium.
This can be seen in the evolution of life, where species become more complex and adaptive over time, as well as in the growth of technology, where innovations allow us to become more efficient and productive. In essence, Negative Entropy is the power that allows open systems to improve and evolve. Therefore Correct answer is option C
Know more about thermodynamics here:
https://brainly.com/question/1368306
#SPJ11